Auto catalyst
An autocatalyst is a cylinder of the elliptical cross-section made from ceramic or metal formed into a fine honeycomb and coated with a solution of chemicals and platinum group metals (PGMs). It is typically mounted inside a stainless steel canister (the whole assembly is called a catalytic converter) and is installed in the exhaust line of a vehicle between the engine and the silencer. Autocatalysts convert over 90% of hydrocarbons, carbon monoxide and oxides of nitrogen from gasoline engines into less harmful carbon dioxide, nitrogen and water vapour.
Autocatalysts also reduce the pollutants in diesel exhaust by converting 90 per cent of hydrocarbons and carbon monoxide and 30 to 40% of particulate into carbon dioxide and water vapour.
The most widespread recycling of PGMs takes place in the autocatalyst sector. It is estimated that 15-20% of the world’s PGM is recovered from spent autocatalysts and that this represents three-fourths of the total secondary PGM supply.
In the North American market, for example, where the use of catalytic converters has been mandated for more than 30 years, recovered metal now accounts for more than half of the platinum required for the manufacture of new catalytic converters.
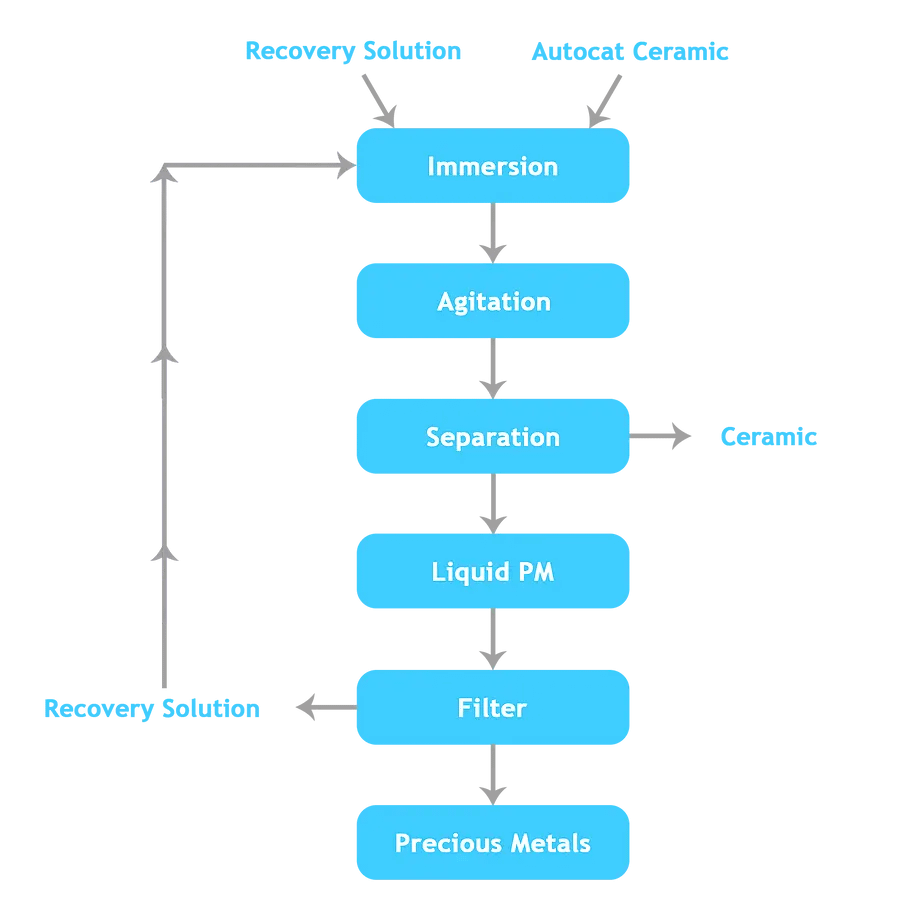
Current Solution
Currently, automotive catalysts are recycled to recover PGMs in both integrated smelter processes and dedicated high-temperature electric furnaces. Pyrometallurgical processing of spent catalysts involves the use of heat to separate the desired metals from unwanted materials by exploiting differences in oxidation potential, melting point, vapour pressure, density and/or miscibility when melted. These smelting and conversion processes can have a highly detrimental impact on the environment, particularly air quality, and are known to release the following hazardous products: